Aseptic Fill & Finish Services for Commercial Products
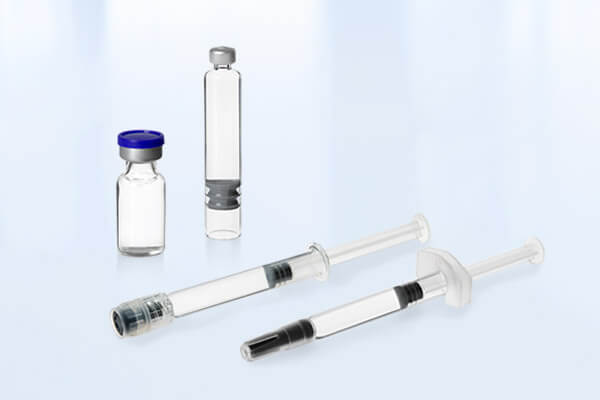
In addition to developing products and scaling them up to our commercial production lines, we also offer aseptic contract filling services for fully developed and approved products. We use the latest state-of-the-art filling equipment from groninger (filling line), SKAN (isolator) and Martin Christ (freeze dryer) with an annual capacity of approx 15 million Ready-To-Use (RTU) primary packaging containers. This includes pre-filled syringes, vials and cartridges in glass and polymer.
Our premises, filling equipment, associated processes and risk assessments already meet the requirements of the new EU GMP Annex 1 today.
Project requests to Thorsten Häfner
E-mail: service@psm-saar.de



Our End-to-End Pharma CDMO Services for Commercial Products
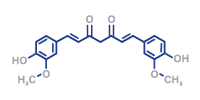
- Manufacturing and compounding of drug substance for example: Biopharmaceuticals, Biosimilars, Small Molecules, Diluents and Ophthalmics
- Aseptic filling, stoppering and closing of pre-fillable syringes, vials and cartridges in different batch sizes up to around 600 liters
- Capabilities for nitrogen overlay / gassing and vacuum options for oxygen sensitive products
- Terminal sterilization
- Analytical services for product release
- Visual inspection, labelling and secondary packaging including GMP storage
- Storage and shipping of products at ambient temperature, cooled at 2°- 8° or frozen down to -75°C.

With the Groninger Flexfill filling system, we can fill batch sizes of up to 600 liters. We offer maximum flexibility in processing ready-to-use syringes up to 50 ml, vials up to 100 ml, and cartridges up to 3 ml in glass or polymer. This line is equipped with peristalitc and rotary piston pumps and various gassing and vacuum options.